The Current Cancer Crisis
Over 1,600 people die each day from cancer in the U.S., according to recent data from the American Cancer Society (1). The 4.8 percent increase in death rate from cancer over the last five years is greater than the 4.2 percent increase in U.S. population growth over the same period.
In this context, it is important to take a close look at both the prevailing and alternative theories of cancer’s origin. Over the next several weeks, we will review the history of and evidence for the somatic mutation theory (SMT) and the mitochondrial metabolic theory (MMT) of cancer.
The Somatic Mutation Theory Persists as the Dominant Explanation for the Origin of Cancer
The prevailing view today is that cancer is a “genetic disease” involving nuclear mutations in oncogenes and tumor suppressor genes (2-4). A typical tumor is thought to contain several so-called “driver gene” mutations that regulate the tumorigenic phenotype (5,6). The nuclear genomic instability seen in nearly all types of tumor cells is considered the primary cause of the cancer’s hallmarks, which include sustained proliferative signaling, evasion of growth suppressors, resistance to cell death, replicative immortality, enhanced vascularization, and activation of invasion and metastasis (2). Somatic mutations, which arise randomly during DNA replication in normal non-cancerous stem cells, are considered the origin of cancer (7).
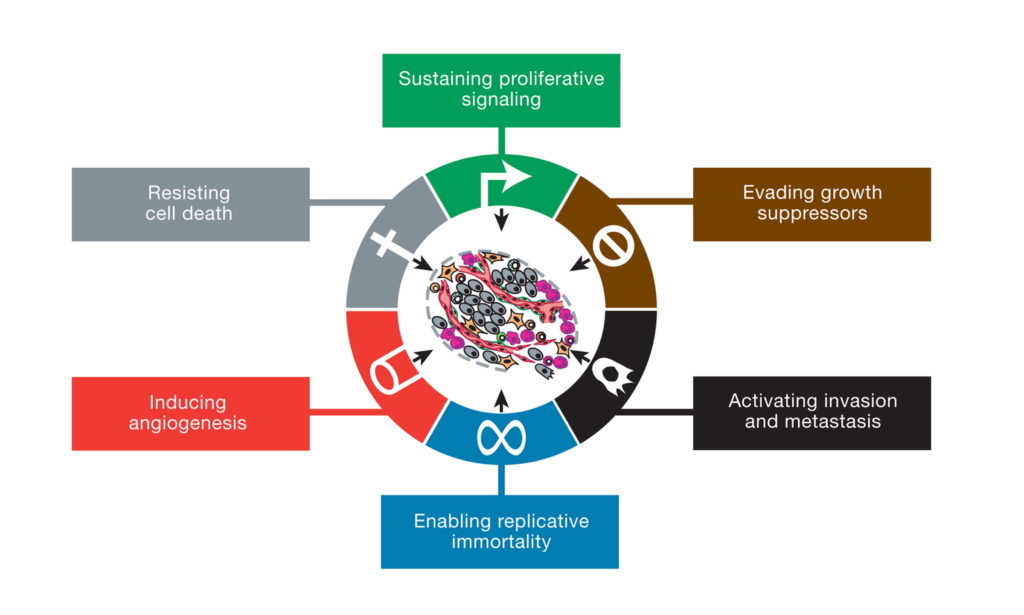
This illustration encompasses the six hallmark capabilities of cancer, originally proposed in 2000 and since substantiated with metabolic underpinnings. From (2).
The somatic mutation theory reigns as the most widely accepted view of the origin of cancer and is the justification for developing personalized genetic therapies or precision medicine for managing the various forms of the disease (4, 8-10). The theory is presented as proven law in most current college textbooks of genetics, biochemistry, and cell biology and is the mainstay of the multi-billion-dollar cancer industry and of the National Cancer Institute (NCI). The NCI website states, “Cancer is a genetic disease—that is, it is caused by changes to genes that control the way our cells function, especially how they grow and divide.” The cancer drug industry and the NIH both consider cancer to be a genetic disease.
The Mitochondrial Metabolic Theory of Cancer
Although the SMT is currently the dominant theory for the origin of cancer, the mitochondrial metabolic theory is emerging as an alternative explanation. Mitochondria are the organelles that produce most cellular energy and are largely responsible for maintaining metabolic homeostasis of the body. The MMT originated with the work of Otto Warburg in the last century and has been resurrected more recently by the work of Seyfried and others (11-17).
The MMT argues that cancer arises primarily from defects in energy production through oxidative phosphorylation (OxPhos) in the mitochondria. OxPhos generates the majority of energy for most cells of the body. Defects in the number, structure, and function of mitochondria will cause cells to gradually replace insufficient respiration with fermentation for energy production, thus initiating the path to neoplasia. Aerobic fermentation of lactic acid, also called the Warburg effect, is recognized as the most common pathological phenotype of cancer. Recent evidence also shows that tumor cells can also use mitochondrial substrate level phosphorylation as another fermentation pathway to compensate for defective respiration. Mitochondrial substrate level phosphorylation is now recognized as the “missing link” in Warburg’s central theory (18). Defective OxPhos with a compensatory reliance on fermentation for energy produces reactive oxygen species (ROS) that are both mutagenic and carcinogenic. According to the MMT, the somatic mutations and all other hallmarks of cancer are considered downstream epiphenomenon for the initial damage to respiration. Hence, the MMT differs from the EMT in placing the origin of the disease in the mitochondria rather than in the nucleus of the cell.
Thomas N. Seyfried is professor of biology at Boston College. He received a doctorate in genetics and biochemistry from the University of Illinois—Urbana-Champaign in 1976. He did his undergraduate work at the University of New England, where he recently received the distinguished Alumni Achievement Award. He also holds a master’s degree in genetics from Illinois State University. Seyfried served with distinction in the United States Army’s 1st Cavalry Division during the Vietnam War and received numerous medals and commendations.
He was a postdoctoral fellow in the Department of Neurology at the Yale University School of Medicine and then served on the faculty as an assistant professor in neurology. Seyfried previously served as chair of the Scientific Advisory Committee for the National Tay-Sachs and Allied Diseases Association. He recently received a Lifetime Achievement Award from the Academy of Complementary and Integrative Medicine and the Uncompromising Science Award from the American College of Nutrition for his work on cancer.
He presently serves on several editorial boards, including those for Nutrition & Metabolism, Neurochemical Research, the Journal of Lipid Research, and ASN Neuro. Seyfried has over 180 peer-reviewed publications and is author of the book “Cancer as a Metabolic Disease: On the Origin, Management, and Prevention of Cancer” (Wiley Press).
References
- 1. Siegel RL, Miller KD, and Jemal A. Cancer statistics, 2018. CA: A Cancer Journal for Clinicians 68.1(2018): 7-30. Available here.
- Hanahan D and Weinberg RA. Hallmarks of cancer: the next generation. Cell 144.5(2011): 646-674. Available here.
- Vogelstein B, Papadopoulos N, Velculescu VE et al. Cancer genome landscapes. Science 339.6127(2013): 1546-1558. Available here.
- Hou JP and Ma J. DawnRank: discovering personalized driver genes in cancer. Genome Medicine 6.7(2014): 56. Available here.
- Iranzo J, Martincorena I, and Koonin EV. Cancer-mutation network and the number and specificity of driver mutations. Proceedings of the National Academy of Sciences of the United States of America 115.26(2018): E6010-E6019. Available here.
- Fearon ER and Vogelstein B. A genetic model for colorectal tumorigenesis. Cell 61.5(1990): 759-767. Available here.
- Tomasetti C and Vogelstein B. Variation in cancer risk among tissues can be explained by the number of stem cell divisions. Science 347.6217(2015): 78-81. Available here.
- Vaux DL. In defense of the somatic mutation theory of cancer. BioEssays 33.5(2011): 341-343. Available here.
- McLeod HL. Cancer pharmacogenomics: early promise, but concerted effort needed. Science 339.6127(2013): 1563-1566. Available here.
- Ju J, Zhu A, and Yuan P. Progress in targeted therapy for breast cancer. Chronic Diseases and Translational Medicine 4.3(2018): 164-175. Available here.
- Seyfried TN. Cancer as a Metabolic Disease: On the Origin, Management and Prevention of Cancer. Hoboken, New Jersey: John Wiley & Sons, Inc., 2012. Available here.
- John AP. Dysfunctional mitochondria, not oxygen insufficiency, cause cancer cells to produce inordinate amounts of lactic acid: the impact of this on the treatment of cancer. Medical Hypotheses 57.4(2001): 429-431. Available here.
- Kim A. Mitochondria in cancer energy metabolism: culprits or bystanders? Toxicological Research 31.4(2015): 323-330. Available here.
- Pelicano H, Zhang W, Liu J et al. Mitochondrial dysfunction in some triple-negative breast cancer cell lines: role of mTOR pathway and therapeutic potential. Breast Cancer Research 16.5(2014): 434. Available here.
- Srinivasan S, Guha M, Dong DW et al. Disruption of cytochrome c oxidase function induces the Warburg effect and metabolic reprogramming. Oncogene 35.12(2016): 1585-1595. Available here.
- Stefano GB and Kream RM. Cancer: mitochondrial origins. Medical Science Monitor 21(2015): 3736-3739. Available here.
- Warburg O. On the origin of cancer cells. Science 123.3191(1956): 309-314. Available here.
- Chinopoulos C and Seyfried TN. Mitochondrial substrate-level phosphorylation as energy source for glioblastoma: review and hypothesis. ASN Neuro 10(2018): 1-27. Available here.
All links accessed Jan. 20, 2019.
Comments on Is Cancer a Genetic or Metabolic Disease? Part 1
This could be the beginning of CrossFit Boxes being recognized as the main prevention centers for Cancer. If the MMT is correct this is also a brighter future for the generation to come : you can avoid cancer! It isn't written in your genes as a death sentence.
There is no doubt that it behooves us all to figure this one out. Because if cancer is truly a metabolic disease, it is then pretty obvious how to more effectively treat/manage it or, better, prevent it in the first place: eat meat, vegetables, nuts and seeds, some fruit, little starch, and no sugar. And, as would be the case for treating other recalcitrant metabolic disorders, if we want to exert maximum downward pressure on the system, I might tweak it to 'Eat meat, some vegetables, certain nuts, little fruit, and NO starch and sugar.' Maybe, for some, even just 'Eat meat.' And if it isn't a metabolic disease--i.e., if the SMT were, in fact, correct, which I don't believe is the case, personally--then we may be stuck with the current horrific set of circumstances from unacceptable incidence rates and persistently poor treatment outcomes.
From up close and personal (viewed from the perspective multiple times with close family members over the last 30 years, rather than from my vantage point as a physician) the standard treatment protocols for cancer--irradiate it and poison it--appear to stem from the position that it's genetic and once it 'happens' all you can do is try to obliterate it somehow. That plan was certainly the case for my sister a few years ago, when she received the diagnosis of highly undifferentiated adenocarcinoma of the lung, stage IV, widely metastatic and inoperable. Left untreated, I'd guess she would have succumbed within a few weeks of diagnosis, as the mets in her mediastinum had encroached on her larynx and esophagus, making it difficult to speak or swallow and those in her brain had already prevented her from being able to think clearly or write her name. Her choice was to 'pull out the treatment stops' and so her oncologist obliged with a course of 30+ packets of irradiation to chest and head and a 6-week platinum combination regimen. After 3 months of misery, several hundred thousand dollars of health care dispensed for hospitals, doctors, radiation, chemo, a DVT, readmission for placement of an IV filter, and another re-admission for a bleeding gastric ulcer, she died. Sadly, I don't think her trajectory is all that unusual.
The 'cure rates' are abysmal for most cancers and their astronomical costs bankrupt families and will surely do the same to the overall health care bank ultimately if the current course is maintained. While there are a handful of treatment success stories--testicular cancer, oat cell cancer of the lung, childhood leukemias, and a few others -- in most instances, the therapy consumes hundreds of thousands of health care dollars, often in exchange for a scant few last months of misery and discomfort. Why does this sad state of affairs go on?
Chercher l'argent!
Right now cancer research dollars are aimed at slicing and dicing the SMT and standard cancer treatment focused on chemical and or mechanical obliteration. Why, when the success rate sucks? Both in research and treatment, if you're being paid to swing a hammer, everything looks like a nail.
A couple notes from Seyfried’s citations…
#2 (Hallmarks of Cancer…) positions reprogramming energy metabolism as an “emerging hallmark” of cancer, citing a series of papers making many of the same (uncontroversial by themselves) observations on which the mitochondrial mutation theory is based. They argue, however, this mitochondrial dysfunction is secondary to other elements of cancer progression, unlike Seyfried’s theory which frames it as primary:
“Altered energy metabolism is proving to be as widespread in cancer cells as many of the other cancer-associated traits that have been accepted as hallmarks of cancer. This realization raises the question of whether deregulating cellular energy metabolism is therefore a core hallmark capability of cancer cells that is as fundamental as the six well-established core hallmarks. In fact, the redirection of energy metabolism is largely orchestrated by proteins that are involved in one way or another in programming the core hallmarks of cancer. When viewed in this way, aerobic glycolysis is simply another phenotype that is programmed by proliferation-inducing oncogenes.”
https://www.cell.com/cell/fulltext/S0092-8674(11)00127-9
This reflects the way the SMT acknowledges observed metabolic defects in cancers - a defect observed in cancers, consistently, but a consequence of cancer progression, not a cause.
Conversely, Seyfried and some of the papers he cites argue mitochondrial defects play a more direct, and even causal, role in cancer progression.
https://www.sciencedirect.com/science/article/pii/S0306987701913350?via%3Dihub
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4751441/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6311572/
Seyfried notes the difference between the MMT and SMT is a question of primacy. The base claim that metabolism is distorted in most cancers is uncontentious. The observation that mitochondrial defects play a role in this metabolic distortion, while possibly a bit more obscure, has clear mechanistic justification. It’s really only the narrowest claim, that the initial damage leading to cancer happens in the mitochondria, not the nucleus, that is contentious.
That said, this narrow claim could have substantial clinical implications.

Is Cancer a Genetic or Metabolic Disease? Part 1
3